Biomedical engineering is constantly moving away from systems that need controlled environments, rigid hardware, or long experimental timelines. Newer approaches are making biological signals easier to capture, manipulate, and study.

A researcher examines engineered cardiac cells grown on a biohybrid substrate, where light-driven stimulation is used to control and study heart tissue behavior. Image used courtesy of UC Irvine
Three recent efforts show that shift in different ways: a system that turns everyday earbuds into cardiac sensors, a light-driven interface that replaces electrodes in heart tissue experiments, and a microfluidic platform that compresses decades of human aging into days.
Turning Earbuds Into Cardiac Sensors
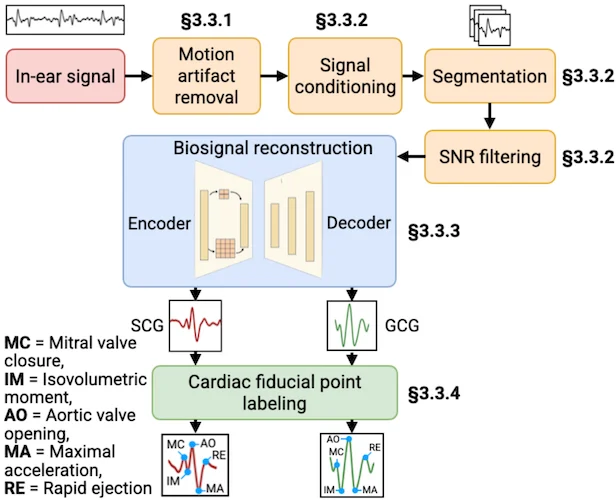
A team at Carnegie Mellon University set out to address a gap that most consumer health devices still can’t touch. While today's smartwatches can track heart rate, they don’t capture the heart's mechanical behavior—specifically, the subtle vibrations tied to valve motion and cardiac timing. Those signals typically require chest-mounted sensors in clinical settings, limiting measurements to short, controlled sessions.

Earbuds repurposed as cardiac sensors capture micro vibrations of the heart. Image used courtesy of Carnegie Mellon University
The team worked with what people already use in everyday life instead of designing new hardware. They repurposed the built-in speaker in earbuds, reversing its function so it acts as a vibration-sensitive transducer. The speaker responds to tiny mechanical signals traveling through the body rather than pushing air to create sound. Those signals are captured at the ear and processed by a machine learning pipeline that reconstructs seismocardiographic and gyrocardiographic waveforms. From there, the system extracts the timing of key cardiac events such as valve opening and closing, shifting the measurement from simple pulse tracking to a more detailed mechanical view of heart function.
The system no longer depends on specialized equipment or clinical environments, and it doesn’t interrupt normal activity. It works when the earbud is in use, effectively turning passive listening time into continuous beneficial monitoring. The team conducted a study with 18 participants to test the approach, comparing earbud-derived signals to those from standard chest-mounted medical sensors. The reconstructed signals closely matched the clinical measurements, with correlations ranging from 0.88 to 0.95. The results confirm that cardiac vibrations propagate through the body in a predictable way and can be reliably detected at the ear.
System pipeline of the "LubDubDecoder". Image used courtesy of Arxiv
The value here isn’t just in the convenience; irregularities in the heart often appear before symptoms manifest. This type of monitoring could identify issues earlier than traditional methods. The team sees applications in tracking conditions such as atrial fibrillation and valve disorders, with next steps focused on scaling validation and refining the system.
Replacing Electrodes With Light
A team at the University of California, Irvine, approached a different limitation: how cardiac tissue is stimulated and studied. Traditional systems rely on metal electrodes to deliver electrical signals, but those electrodes don’t match the softness and constant motion of heart tissue. Over time, they can introduce damage, contamination, or measurement artifacts. More advanced approaches, such as optogenetics, improve control but require genetic modification, limiting their practicality.
The team developed a biohybrid interface that replaces electrodes entirely. The system combines layers of optoelectronic polymers with living cardiac cells, creating a flexible platform that converts light into electrical stimulation.

Light-triggered electrical and mechanical stimulation. Image used courtesy of Cell Biomaterials
The polymer generates photocurrents at its surface when exposed to pulsed visible light. These currents interact with the surrounding cell environment, triggering electrical activity in the cardiomyocytes and causing them to contract. Because the mechanism is material-based, it works with unmodified cells, avoiding the constraints of genetic approaches.
This design solves a core problem by aligning the interface with the biology. The material is mechanically compliant, so it moves with the tissue rather than against it, and the stimulation is localized and controlled without relying on rigid components. The system was tested using cultured cardiac cells arranged to mimic the structure of native heart tissue. The cells began contracting in sync with the input pulses under light stimulation, effectively reproducing a controlled heartbeat. The setup also allowed researchers to directly observe the mechanical response of the tissue, linking electrical stimulation to physical behavior in a single system.
The team is positioning the system for drug screening and disease modeling. They envision implantable cardiac patches that use light-based pacing, with ongoing work to adapt the system to wavelengths that penetrate deeper into tissue.
Recreating Aging on a Chip
Researchers at the University of California, Berkeley, focused on a broader bottleneck: studying human aging in a way that is both fast and biologically accurate. Drug development relies heavily on animal models and long-term studies, yet most therapies fail in human trials. A major reason is that existing systems don’t capture how human tissues actually age.
To address this, the team built a microfluidic organ-on-a-chip platform that recreates aging in human tissue. The device contains interconnected chambers of fat and liver cells derived from human stem cells linked by channels that simulate blood flow between organs.
The system is based on the key insight that aging is driven in part by circulating factors in the blood. By exposing the tissue to serum from older donors, the platform induces aging-related changes directly within the cells. Exposure to younger serum produces the opposite effect, maintaining more youthful behavior. Because the chambers are connected, signals from one tissue can influence another, replicating how aging propagates across organs.

UC Berkeley’s organ-on-a-chip platform uses interconnected chambers of fat and liver cells to replicate human biological signaling. Image used courtesy of UC Berkeley
What makes the platform stand out is speed. Within four days, the system reproduced changes that normally take decades in humans. Cells exposed to older serum showed inflammation, metabolic dysfunction, senescence, and DNA damage. The team validated the model by comparing gene expression patterns from the chip with those from large human datasets. A machine learning model trained on those datasets predicted the biological age of chip-grown tissues with 90-97% accuracy, confirming that the system closely mirrors real aging.
The platform also exposed patterns that traditional models usually miss. Aging in the fat tissue did not stay contained there. Once those changes set in, the liver began showing damage as well. The system also picked up clear differences in how aging played out across male and female samples. Beyond observation, the team used the platform to test interventions. Some treatments, such as oxytocin, improved metabolic function and reduced inflammation, while other widely used therapies, like rapamycin, showed little benefit.
These results highlight the system’s ability to rapidly evaluate therapeutic effectiveness in a human-relevant context. The team has identified new biomarkers of aging and is advancing toward commercialization, positioning the platform as a tool to accelerate drug discovery and improve the reliability of preclinical testing.
A Shift Toward More Practical Biology
Each of these systems removes a different constraint. Together, they point toward a change in how biological systems are studied and used. The shift from constrained, high-friction systems to accessible, scalable ones is where real progress is happening.
-
Tel
+86 180 2549 2789 -
Wechat

 BOM
BOM Cart()
Cart() English
English Russia
Russia Korean
Korean
